by Alexander Geragotellis
Background:
Burns are a global public health challenge and claim the lives of ±180 000 people each year [1]. The vast majority (±90-95%) of these deaths occur in LMICs and disproportionately afflict African children under the age of 5 years, who carry double the incidence of burn deaths compared to children under the age of 5 years worldwide [1]. Despite the extensive catalogue of burn wound coverage options, skin repair could still be defective owing to the complexity of the healing process. Cell-mediated treatment achieved through bioengineered constructs may provide alternative and more effective avenues to resolve burn wounds. Hydrogels are valuable tools in tissue engineering and regenerative medicine for stabilising tissue, and for delivering growth factors, bioactive ingredients and stem cells. The polymers used to make hydrogels can be natural [2], synthetic [3] or hybridised using both polymer types to infuse the desirable properties into one construct. The group who published the current research paper have previously reported on the advantageous properties of the poly(ethylene-glycol)-fibrin hybrid hydrogels [4]. Figure 1 offers a resource with further clarity on relevant tissue engineering terminology.
Aim:
The current study [5] aimed to evaluate the burn-wound healing potential of a bio-engineered 3D PEGylated-fibrin (P-fibrin) hybrid hydrogel as a scaffold to deliver adipocyte derived stem-cell (ASCs) using a rat model. The synthesis scheme has been summarised in Figure 2.

Figure 1 – Glossary of useful terminology to guide interpretation of the paper and results.
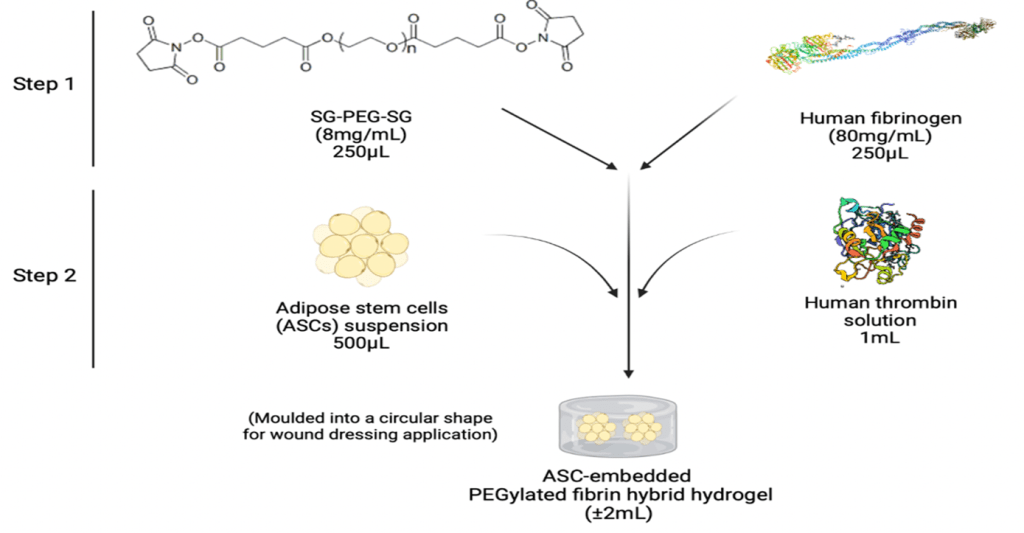
Figure 2 – Synthesis scheme for the ASC-embedded P-fibrin hybrid hydrogels used in the study [5]. ASCs were embedded within the hydrogels to try further augment the pro-regenerative activity of the P-fibrin hydrogels.
Objectives:
Methods used to achieve objectives have been summarised in Figure 3. By comparing burns wounds (i) untreated, or treated with either (ii) P-fibrin or (iii) P-fibrin with embedded ASCs between 7-21 days post- burn injury, authors set out to determine:
- Whether or not the application of the gels negatively affected burn wound closure.
- Characterise wound coverage and wound integration of the hydrogels using light and fluorescent microscopy.
- Explore angiogenic potential via immunohistochemical analysis of burn wounds to quantify blood vessels.
- Evaluate macrophage activation status of regenerating wounds, and how it is impacted by hydrogel application using immunohistochemistry.
- Assess collagen deposition and organisation 21 days post-injury to understand longer-term recovery phenotype.
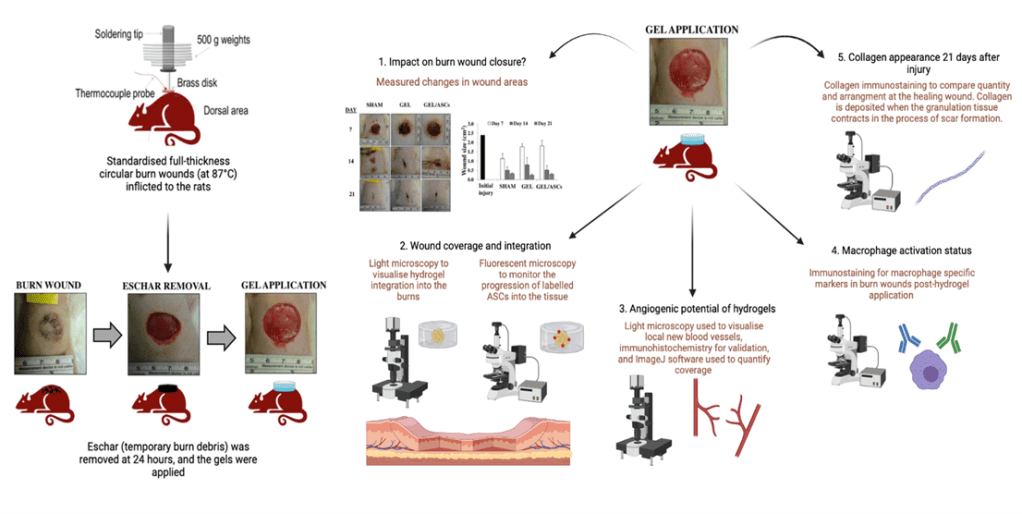
Figure 3 – Rat burn model, adapted from Chung et. al [5], and accompanying methodologies used to achieve the objectives of the study.
Take home messages:
- The major value of using a P-fibrin hydrogel appears to be with the increased cellularity of the granulation tissue at one week post-burn, suggesting an increase in tissue regeneration capability. The increased cellularity was similar to that observed when the P-fibrin was embedded with ASCs, indicating that the scaffold itself was the responsible factor and that the ASCs had little effect.
- Angiogenesis is central process in tissue regeneration. The major value of embedding ASCs within the P-fibrin scaffolds was the bolstered neovascularisation observed at 7 days post-burn, which was significantly superior to the small increase observed with the P-fibrin only group. This would support the authors’ supplementary findings that suggest a role of the ASC secretome upregulating pro-angiogenic genes [5]. Although not fully studied, it is likely that the ASCs influence the population diversity of regenerative cells recruited to the burn wound to influence this superior angiogenic response.
- To this end, there is some evidence in the present study to suggest that embedded ASCs can inflict a local tolerising pro-regenerative phenotype. Specifically, wounds treated with P-Fibrin embedded with ASCs showed a drastically greater coverage of the M2 immunosuppressive, pro-regenerative macrophages than when the wounds were treated with P-Fibrin gel alone.
- It is encouraging that the wound thickness, collagen deposition and organisation between the studied hydrogel groups did not vary significantly, because this suggests that the hydrogels only accelerate but do not change the orchestrated sequence of the in-vivo regenerative response to full-thickness burns.
- P-fibrin hydrogels hold great promise for burn wound healing applications, especially when conjugated with biological agents. Infection control is a crucial consideration in the burn wound healing process, and was not explored in the present study [5]. Interestingly, a recent study found that both infection control and neovascularisation post-burn can be enhanced through sequential delivery of P-fibrin embedded silver sulfadiazine loaded chitosan microspheres followed by P-fibrin embedded ASCs [6]. Future studies will likely focus on further characterising similar functional hydrogel constructs prior to progressing to more advanced trials.
References:
- World Health Organisation. 2018. Burns. Online: https://www.who.int/news-room/fact-sheets/detail/burns
- Janmey PA, Winer JP, Weisel JW. Fibrin gels and their clinical and bioengineering applications. J R Soc Interface. 2009;6(30):1-10.
- Overby RJ, Feldman DS. Influence of Poly (Ethylene Glycol) End Groups on Poly (Ethylene Glycol)-Albumin System Properties as a Potential Degradable Tissue Scaffold. Journal of Functional Biomaterials. 2019;10:1.
- Nam SY, Chung E, Suggs LJ, Emelianov SY. Combined ultrasound and photoacoustic imaging to noninvasively assess burn injury and selectively monitor a regenerative tissue-engineered construct. Tissue Eng Part C Methods. 2015;21(6):557-566.
- Chung E, Rybalko VY, Hsieh PL, Leal SL, Samano MA, Willauer AN, et al. Fibrin-based stem cell containing scaffold improves the dynamics of burn wound healing. Wound Repair Regen 2016;24:810–9.
- Banerjee J, Seetharaman S, Wrice NL, Christy RJ, Natesan S. Delivery of silver sulfadiazine and adipose derived stem cells using fibrin hydrogel improves infected burn wound regeneration. PLoS One. 2019;14(6):e0217965.
Leave a comment