by Nimra Khan
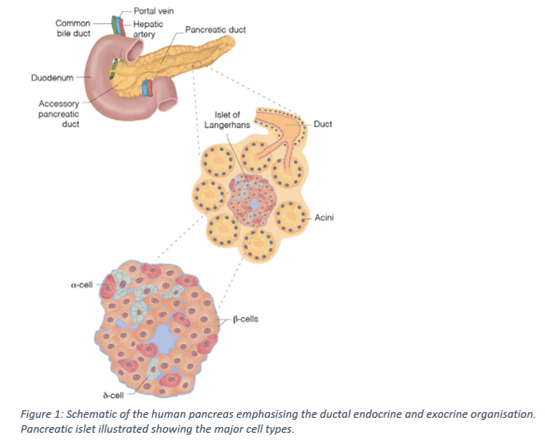
The global disease burden of diabetes is 536.6 million people worldwide. Diabetes can be divided into Type 1 and Type 2 diabetes mellitus. Type 1 diabetes (T1D) is an autoimmune disease that destroys insulin-producing Beta (β) cells in the pancreatic islets (Figure 1). Insulin is a hormone made in your pancreas; a gland located behind your stomach. Insulin allows your body to use glucose for energy. People with T1D rely on daily insulin injections to replace what would have been produced by their β cells. Complications related to diabetes includes an increased risk of cardiovascular disease, kidney failure, heart attack, stroke, lower limb amputation, nerve damage and vision impairment. Current strategy of replacing the damaged β-cell mass involves whole pancreas transplantation. Because of a shortage of organ donors and problems associated with immunosuppressive drugs, transplantation strategies have limited clinical use. Therefore, looking at the possibility of regenerating β-cells in the pancreases is of interest.
Intriguingly, the human epigenetics research team from Monash University in Melbourne, Australia was able to “reawaken” stem-like cells in the pancreas and re-establish insulin expression from those cells using a drug approved by the US FDA. The research team published their findings in the article “Inhibition of pancreatic EZH2 restores progenitor insulin in T1D donor” in Signal Transduction and Targeted Therapy.
Experimental evidence shows inherent plasticity (ability to change function) of pancreatic cells, this fuelled interest in the potential regeneration of β-cells from other pancreatic cells (Figure 1). This potential regeneration of β-cells was previously demonstrated with the discovery of Alpha (α) cell plasticity and the ability of ductal and α-cells to convert into insulin-producing cells in the pancreases. It is also important to understand that the pancreas showed classic silencing of β-cell progenitor (ancestral) genes with barely detectable insulin transcript. This default silencing occurs due to EZH2 gene’s methyltransferase protein activity on H3K27me3. H3K27me3 is an epigenetic modification to the DNA packaging protein called Histone 3, this moditification functions to silence the insulin gene using EZH2 protein.
Therefore, the objective of this study was to investigate the effect of the drug GSK126 on the specific regulation of the H3K27me3 activity by EZH2 protein that is considered responsible for default suppression (transcriptional gene silencing). The overcoming of this suppression is critical to the progenitor cell’s developmental programme by enabling them to differentiate into functioning insulin secreting β-cells.
Isolated cells derived from the pancreas (Figure 2) were stimulated with GSK126 – a specific pharmacological inhibitor of EZH2 gene’s methyltransferase protein. Results showed that the drug effectively reduced h3k27me3 content, thereby influencing beta cell plasticity and promoting insulin gene expression (Figure 3). GSK126 also reinstated Insulin gene expression despite absolute β-cell destruction, dramatically influencing insulin expression in diabetic donor cells with damaged pancreatic islets. Treating the ductal cells with the drug GSK126 caused them to functionally resemble β cells and to produce insulin. These results showed a promising future for T1D treatment.
The main advantage of this method of treatment over beta cell replacement includes rapid insulin restoration requiring several days of drug treatment only as opposed to other protocols that can take several months. This novel research approach would allow insulin-producing β cells that are destroyed in T1D patients to be replaced with new insulin-generating cells. An important step towards developing new therapies.
Concluding remarks and future perspectives
Diabetes affects millions of people worldwide, this research is a starting point towards aiming to improve the quality of life of individuals with diabetes by reducing the need for insulin injections, dietary and activity restrictions, and importantly eliminate the need for pancreas transplants in chronic sufferers. These scientists discovered a way to “awaken” stem cells in the pancreas and harness their ability to express insulin.
Why is this discovery important? Because this provides a potential for treatment whereby using the patient’s own pancreas cells we can turn on genes which promote the production of insulin and will ultimately reduce the dependency on insulin injections (up to 100 per month) and eliminate the need for pancreas transplants.


References
Al-Hasani, K., Khurana, I., Mariana, L., Loudovaris, T., Maxwell, S., Harikrishnan, K. N., Okabe, J., Cooper, M. E., & El-Osta, A. (2022). Inhibition of pancreatic EZH2 restores progenitor insulin in T1D donor. Signal transduction and targeted therapy, 7(1), 248. https://doi.org/10.1038/s41392-022-01034-7
Healthline. (2019). 10 Facts About Type 2 Diabetes and Insulin: Types, Risks, and More. [online] Available at: https://www.healthline.com/health/type-2-diabetes/insulin-facts-to-know#habits [Accessed 16 Sep. 2022].
Leave a comment