by Imraan Dixon
Imagine the things your body is capable of. Imagine even the little things like stretching, scratching that itch, talking, and eating. Now, imagine not being able to do any of that, each ability you’ve had for as long as you can remember slowly eroding away. Every little thing we may take for granted like being able to swallow, being able to speak, or just being able to move our bodies is a gift that a person with amyotrophic lateral sclerosis (ALS) soon loses.
ALS is a neurodegenerative disease characterised by the progressive death of motor neurons in the spine and the brain that control voluntary muscle movements. The death of these neurons leads to the subsequent death of muscle cells. The loss of motor neurons spreads to other parts of the body leading to the inability to move, eat and swallow, and speak. Eventually, this fatally leads to the muscle and nerve cells of the diaphragm – the most important muscle involved in breathing – also dying. Now, symptoms and disease progression may vary, but ultimately one’s life and loved ones are affected severely.
There is unfortunately no current cure for ALS. All that can be done right now is supportive care that can potentially increase life expectancy but at the cost of quality of life. The difficulty in finding effective long-term treatments is due to our lack of understanding of how this disease occurs. There are a multitude of genetic and environmental factors that play a role in ALS, so the picture is quite complex. One fundamental question underpinning ALS pathogenesis is:
“Why do those neurons die?”
Firstly, cell death is divided broadly into two categories. We have accidental cell death that is uncontrolled and typically caused by direct chemical or physical injury. Then, we have regulated cell death (RCD) that is controlled and regulated by our own cells. One major kind of RCD is “apoptosis” which is typically triggered by death ligands (molecules that signal cells to undergo RCD) and a caspase cascade (a chain reaction of caspase molecules that result in apoptosis). It
was originally thought that motor neuron death was caused by apoptosis, but inhibiting it did not protect neurons in ALS. Then, “necroptosis” was suggested as a mechanism. It’s another form of RCD that’s typically caspase-independent and driven by inflammation. However, if we get rid of MLKL – a critical component of necroptotic signalling – we find no changes in neuronal death.
A group of researchers in Australia aimed to find a mechanism underlying neuron death in ALS. That led them to “ferroptosis”. Ferroptosis is a relatively newer discovery and also a form of RCD that is iron-dependent and caspase-independent. Ferroptosis is typically caused by iron dysregulation and glutathione peroxidase 4 (GPX4) depletion that result in lipid peroxidation and cellular membrane damage (lipids are a major component of cell membranes). GPX4 is a major regulator of ferroptosis as it is acts as a defense against lipid peroxides, preventing widespread cellular damage. It does require glutathione (GSH), an antioxidant, in order to do so. See, as it turns out, accumulated iron, lipid peroxidation, and glutathione depletion were found in spinal cord and brain tissue of affected ALS individuals. See the link here?
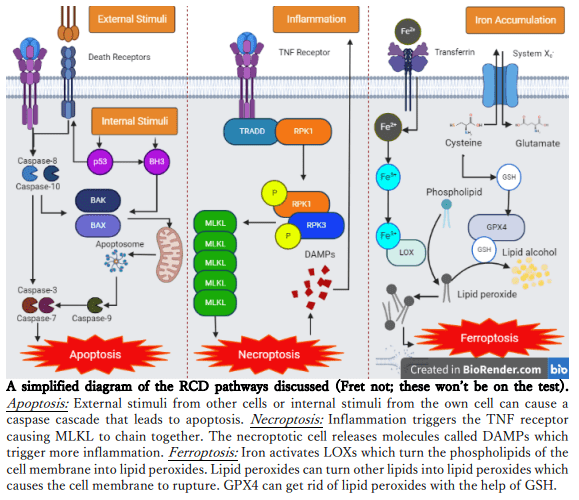
To test if ferroptosis was indeed the cause of cell death in ALS, the researchers tested the effect of GPX4 on neurons from ALS mice. The result? ALS mice had lower levels of GPX4, and iron levels were dysregulated as expected. Overexpressing GPX4 in these mice showed a positive effect. There was a decrease in lipid peroxidation, therefore cell death was decreased, and disease onset was delayed thereby extending the mice’s lifespan. Additionally motor function was somewhat preserved although disease progressed as usual upon onset.
Does it really amount to much though?
These might not seem like groundbreaking discoveries. After all, the ALS mice still displayed neurodegeneration, indicating that we’re yet to uncover the full picture, let alone find a suitable long-term treatment. But we’ve made progress. Highlighting ferroptosis’s role in ALS opens up new avenues for treatments and discovery. Just from the researcher’s suggestions alone, perhaps selenium (a component of GPX4) or GSH treatments may be useful; iron chelation to remove excess iron might also hamper degeneration. Just knowing that ferroptosis exists and is involved in ALS gives us the power to target it in the future. Perhaps this is just the beginning. Ferroptosis is slowly being linked to other neurodegenerative diseases. Understanding it and how to influence it has the possibility of building the foundation upon which we can treat other diseases as well. Every step we take towards learning more and more about the diseases that plague us is a step towards building a world where people don’t have to suffer from them.
Reference
Wang, T., Tomas, D., Perera, N.D., Cuic, B., Luikinga, S., Viden, A., Barton, S.K., McLean, C.A., Samson, A.L., Southon, A. and Bush, A.I., 2022. Ferroptosis mediates selective motor neuron death in amyotrophic lateral sclerosis. Cell Death &
Differentiation, 29(6), pp.1187-1198
Leave a comment