by Kelly-Robyn Singh
Lynch syndrome is an autosomal dominantly inherited cancer syndrome which predisposes individuals to a wide range of cancers, most commonly, cancer of the colon and/or rectum (colorectal cancer- CRC). It presents as colonic adenomas (benign epithelial tissue tumor) at an early age of onset that is averagely seen in other cancers. Lynch syndrome is the most common type of hereditary colon cancer, and it is estimated that approximately 1 in every 300 people carry mutations which are associated with Lynch syndrome. These associated mutations are found in the mismatch repair (MMR) genes, MLH1, MSH2, MSH6 or PMS2. Consider the following analogy: the MMR genes encode the MMR machinery, which we can think of as the police-force of the genetic code, in the sense that they recognize inconsistencies in the genetic code which may have arisen during DNA synthesis, and they repair it to restore genetic stability, much like a policeman/woman would in the instance of crime. In a society without the police to maintain law and order, there would be chaos, much like there is when the mutated MMR genes cause non-functional MMR and subsequently, deleterious mutations are allowed to accumulate in the genetic code, uninterrupted.
The question is, could mutations in some of these mismatch repair genes have a more severe effect than others? Is colonic surveillance and removal of pre-cancerous adenomas an effective strategy to manage CRC risk and incidence? The paper by Møller et al. aims to investigate the effects of surveillance in individuals carrying Lynch Syndrome associated mutations and their risk of cancer in more effective and accurate estimates than the usual risk estimation approaches which mainly involve retrospective studies.
To accomplish this, a cohort of patients carrying mutations in the mismatch repair (MMR) genes, MLH1, MSH2, MSH6 and PMS2 were studied and monitored on an Oracle regional database, with the aim to correlate data across categories like age, sex and mutated gene as the data develops to identify significant trends.
The findings were that 314 patients had developed cancer out of 1942 mutation carriers with no prior history of cancer. The cancers were found were mostly CRC, followed by endometrial and ovarian. Interestingly, carriers with mutations in the MLH1 and MSH2 had an earlier age of onset (25 years) as compared to that of MSH6 and PMS2 carriers (40 years). The cumulative cancer incidence rates at 70 years (patient age)for each gene as well as ten-year crude survival can be found below.
Table 1: Cumulative cancer incidences for each MMR gene and ten-year crude survivals for their respective cancers
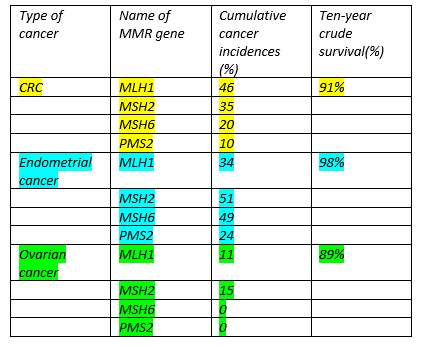

Figure 1: Cumulative incidences by age and mutated MMR gene for any cancer
These findings conclude that there are varied penetrance and expression patterns of each of the mismatch repair genes, with that of MLH1 and MSH2 being higher and therefore causing an earlier age of onset as well as incidence. The results of this paper conclude that colonoscopic surveillance did not significantly decrease the incidence of CRC, but it did result in a lower mortality rate. The early detection of pre-cancerous adenomas is therefore imperative to prevention and the prolonging of life.
References:
Møller P, Seppälä T, Bernstein I, Holinski-Feder E, Sala P, Evans DG, Lindblom A, Macrae F, Blanco I, Sijmons R, Jeffries J, Vasen H, Burn J, Nakken S, Hovig E, Rødland EA, Tharmaratnam K, de Vos Tot Nederveen Cappel WH, Hill J, Wijnen J, Green K, Lalloo F, Sunde L, Mints M, Bertario L, Pineda M, Navarro M, Morak M, Renkonen-Sinisalo L, Frayling IM, Plazzer JP, Pylvanainen K, Sampson JR, Capella G, Mecklin JP, Möslein G; Mallorca Group (http://mallorca-group.eu). Cancer incidence and survival in Lynch syndrome patients receiving colonoscopic and gynaecological surveillance: first report from the prospective Lynch syndrome database. Gut. 2017 Mar;66(3):464-472. doi: 10.1136/gutjnl-2015-309675. Epub 2015 Dec 9. PMID: 26657901; PMCID: PMC5534760.
Leave a comment