by Moses Malebana
Following the outbreak of SARS-COV2 in 2019, there has been a massive global effort to find the cure for the disease. In the midst of all the efforts, it was announced in 2020 by the press that a “cure” for COVID-19 has been found, in an African country, Madagascar. The “cure” was said to be from a herb, Artermessia annua, commonly known as Umhlonyana or Lengana in South Africa. Despite the claims, the remedy could not be accepted by the scientific community and WHO due to the lack of scientific evidence behind the claims. Since then, a little has been said about the remedy which left many people wondering what has happened to our Africa founded “cure”. In this article, we are looking to delve into the available and recent scientific literature to answer a very important question, what is science saying about Umhlonyana?
In-vitro studies
In a recent publication by Zhou Y et al, the scientists aimed to determine the in-vitro efficacy of Artemessia annua extracts on SARS-CoV-2. To determine the efficacy, the researchers cultured SARS-CoV2 in the plates and treated with varying concentration of the A. annua extracts then measured the effects of the extracts on the viral growth. In addition, they treated normal lung epithelial cells and measured the toxicity of the extracts on normal/non-infected cells.
The results showed that the A. annua extracts are able to potently inhibit the growth of SARS-CoV2 at the concentrations between 140 ug/ml to 260 ug/ml (fig.1) (1). In addition, the cytotoxic concentration which is the concentration at which the extracts are toxic to normal cells is 7-10 folds higher than the effective concentration (1). This is very promising as it suggests that the extracts have the potential to inhibit the growth of the virus with minimal risk of side effects.

Figure 1: Results from Zhou et al, 2021 showing the effect of A. annua extracts on SARS-CoV2 growth and normal cell viability.
The second group of scientists in Germany also looked into the effects of Artemessia annua extracts on SARS-CoV2. The interesting thing about these experiments is that they made use of the actual Covid-Organics herbs from Madagascar and tested its effects on the SARS-CoV2 growth in vitro. The experiments they did are colony formation assay, which allows one to visually observe the effects of the extracts on viral growth in culture.
The results clearly showed a significant inhibition of SARS-CoV2 growth even when the extract were diluted to 8.3% of the total concentration (fig.2) (2). The researchers went further to compare the effects of other Artemessia species from different countries on SARS-Cov2 growth found that they were also able to inhibit the viral growth (2).
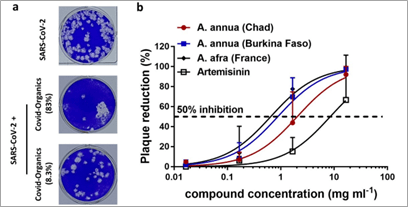
Figure 2 Colony formation assay results of SARS-CoV2 infected cells treated with Covid-Organics (8.3%)
Lastly, the efficacy of A. annua extracts (Artemisinin) was tested against the new SARS-CoV2 variants. This was very important since the emergence of new variants confers resistance to current modes of treatments and preventions. The experiments were similar to the above experiments but this time including the new variants such as variants, B1.1.7 (alpha variant) and B1.351 (beta variant). Interestingly, the extracts were prepared by boiling the Artemessia annua leaves in hot water which is a common and simple method that many people use to prepare the extracts in their homes. As shown on the table, Artemisinin retained its potency across both variants compared to the wild-type virus (fig. 3) (3). These results suggest that not only do A. annua extracts have potential to treat SARS-CoV2, but the extracts might not even need any complicated facilities or equipment to prepare making them very cost-effective and easy forms of treatment.
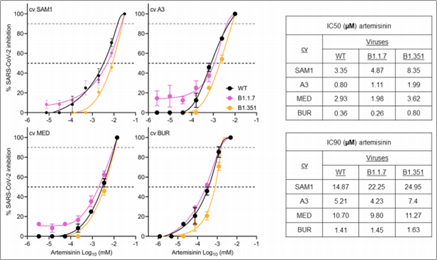
Figure 3| Proliferation assay results showing efficacy of A. annua extracts on different variants.
Clinical trials
It is very clear that Artemessia annua herbal extracts (Umhlonyana) are showing promising results as a potential treatment for COVID-19. This brings out an important question; Can we infer from these results that the herb is a cure for COVID-19? It is important to note that in-vitro experiments are performed without taking into considerations the complex interactions between the human body and the potential drug being tested. As a results, more often that not, potential drugs that show promising results in vitro may not show the same effects when administered to patients.
A. annua extracts did enter into phase II clinical trials to test for their efficacy and safety in humans which were eventually terminated (4). We hope to hear the rationale behind the termination of the clinical trial soon.
References
- Zhou Y, Gilmore K, Ramirez S, Settels E, Gammeltoft K, Pham L et al. In vitro efficacy of artemisinin-based treatments against SARS-CoV-2. Scientific Reports. 2021;11.
- Chuanxiong N, Jakob T, Sooyeon M, Rainer H, Kerry G, Benedikt B. K, Peter H. In vitro efficacy of Artemisia extracts against SARS-CoV-2. Scientific Reports. 2021.
- Nair M, Huang Y, Fidock D, Polyak S, Wagoner J, Towler M et al. Artemisia annua L. extracts inhibit the in vitro replication of SARS-CoV-2 and two of its variants. Journal of Ethnopharmacology. 2021;274:114016.
- Camostat and Artemisia Annua vs Placebo in COVID-19 Outpatients – Full Text View – ClinicalTrials.gov. Clinicaltrials.gov. 2021. Available from: https://clinicaltrials.gov/ct2/show/study/NCT04530617
Leave a comment